pH Levels in the Human Body
Introduction
The human body functions optimally within a narrow range of chemical conditions. Among the most important of these is the pH level, which measures the acidity or alkalinity of body fluids. The term pH stands for “potential of hydrogen” and reflects the concentration of hydrogen ions (H⁺) in a solution. Maintaining a stable pH is essential for normal cellular function, enzymatic activity, and overall metabolic balance. Even slight deviations from the normal pH range can disrupt biological processes and lead to severe health consequences.
Understanding pH
The pH scale ranges from 0 to 14, where:
- A pH of 7 is neutral,
- A pH below 7 is acidic, and
- A pH above 7 is alkaline (basic).
The pH scale is logarithmic, meaning each whole number represents a tenfold difference in hydrogen ion concentration. For example, a solution with a pH of 6 is ten times more acidic than one with a pH of 7.
In the human body, different fluids have different pH values, each optimized for specific functions.
Normal pH Ranges in the Human Body
| Body Fluid | Normal pH Range | Physiological Importance |
| Blood (arterial) | 7.35 – 7.45 | Critical for enzyme and metabolic activity |
| Venous blood | 7.31 – 7.41 | Slightly more acidic due to CO₂ transport |
| Urine | 4.5 – 8.0 | Varies to regulate acid–base balance |
| Gastric juice | 1.5 – 3.5 | Highly acidic for digestion and killing microbes |
| Saliva | 6.2 – 7.6 | Helps neutralize acids in the mouth |
| Cerebrospinal fluid (CSF) | 7.32 – 7.35 | Maintains optimal conditions for neural function |
| Pancreatic fluid | 7.8 – 8.0 | Alkaline to neutralize stomach acid entering the intestine |
The blood pH is particularly important because most enzymes and metabolic processes function effectively only within this narrow range.
Mechanisms Maintaining pH Balance
The body tightly regulates pH through several buffering systems and physiological processes involving the respiratory system, renal system, and chemical buffers.
1. Chemical Buffer Systems
These act within seconds to resist changes in pH by binding or releasing hydrogen ions (H⁺).
The major buffer systems include:
- Bicarbonate Buffer System:
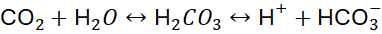
This system maintains blood pH by balancing carbonic acid (H₂CO₃) and bicarbonate (HCO₃⁻).
- Phosphate Buffer System: Important in intracellular and renal fluid regulation.
- Protein Buffer System: Proteins like hemoglobin can bind or release H⁺ to stabilize pH.
2. Respiratory Regulation
The respiratory system controls blood pH by regulating carbon dioxide (CO₂) levels:
- Increased CO₂ → forms more carbonic acid → decreases pH (more acidic)
- Decreased CO₂ → reduces carbonic acid → increases pH (more alkaline)
Breathing rate can change within minutes to compensate for pH imbalances.
3. Renal (Kidney) Regulation
The kidneys provide long-term pH balance by:
- Excreting hydrogen ions (H⁺) into urine
- Reabsorbing bicarbonate ions (HCO₃⁻) into the blood
This process takes hours to days but provides the most precise control of blood pH.
Disorders of pH Imbalance
If pH regulation fails, the body experiences acidosis or alkalosis, which can be life-threatening.
1. Acidosis (pH < 7.35)
- Respiratory acidosis: Caused by CO₂ retention due to hypoventilation (e.g., lung disease).
- Metabolic acidosis: Caused by loss of bicarbonate or buildup of acids (e.g., diabetic ketoacidosis, renal failure).
Symptoms: Fatigue, confusion, headache, rapid breathing.
2. Alkalosis (pH > 7.45)
- Respiratory alkalosis: Results from excessive CO₂ loss due to hyperventilation.
- Metabolic alkalosis: Results from loss of acid (e.g., vomiting) or excess bicarbonate intake.
Symptoms: Muscle twitching, lightheadedness, irritability.
Both conditions require immediate correction to prevent enzyme dysfunction and cellular injury.
Importance of pH in Physiology
- Enzyme Activity:
Enzymes work best at specific pH levels. For example, pepsin in the stomach functions optimally at pH 2, while amylase in saliva works best near pH 7. - Oxygen Transport:
Blood pH affects hemoglobin’s ability to bind and release oxygen — known as the Bohr effect. Lower pH reduces oxygen affinity, allowing more oxygen delivery to tissues. - Electrolyte Balance:
Changes in pH can alter the distribution of ions like potassium (K⁺) and calcium (Ca²⁺), affecting nerve conduction and muscle contraction. - Cell Membrane Function:
pH influences membrane permeability and the function of transport proteins.
Homeostasis of pH
Maintaining pH homeostasis is critical for life. The combined actions of buffers, respiration, and renal function form a multi-layered defense system that prevents dangerous fluctuations. For instance, during intense exercise, the body produces lactic acid, which temporarily lowers pH. The respiratory system responds by increasing breathing rate to expel CO₂, while the kidneys later excrete excess acids to restore balance.
Summary
The human body’s pH balance is a finely tuned system essential for maintaining health and sustaining life. Blood and other body fluids must remain within specific pH ranges to ensure proper enzyme activity, metabolic reactions, and physiological stability. The coordinated efforts of chemical buffers, respiratory control, and renal regulation work continuously to stabilize pH levels. Disruptions in this balance, whether toward acidosis or alkalosis, can have severe consequences, underscoring the importance of homeostatic mechanisms in maintaining the body’s internal environment. Understanding pH regulation not only provides insight into normal physiology but also offers a foundation for diagnosing and treating many medical conditions.
